|
5/28/2023 0 Comments Srdx hda19 tpl
In addition to this overview of ETT structure, the HSQC NMR probes chemical shifts of protein amide-NH bonds in response to the presence of ligand ( Meyer and Peters, 2003). Interestingly, the spectrum also shows dispersed peaks flanking the signal-dense region indicating that there is nevertheless some propensity to form secondary structure, particularly with a helical character ( Figure 1b). The HSQC spectrum, recorded at 5 ☌, shows a prominent signal-dense region consistent with the ES domain being largely intrinsically disordered. Therefore, to test whether ETT binds IAA, we carried out heteronuclear single quantum coherence (HSQC) nuclear magnetic resonance (NMR) experiments using 15N-labelled ES 388-594 protein. The sensitivity of ETT-TF interactions to IAA suggests a direct effect of the IAA molecule on the ETT protein. A protein fragment containing 207 amino acids of the ES domain, ES 388-594, sufficient for mediating IAA-sensitivity in ETT-protein interactions, was produced recombinantly and shown to be intrinsically disordered ( Simonini et al., 2018). The region responsible for IAA-sensitivity is situated within the C-terminal part of ETT, known as the ETT-Specific (ES) domain. Under high auxin conditions, ETT binds auxin leading to dissociation of TPL/TPR-HDA19 and de-repression of ETT targets. In conditions of low auxin levels, ETT interacts with co-repressors of the TOPLESS/TOPLESS-RELATED (TPL/TPR) family to keep chromatin at ETT target loci in a repressed state through HDA19-mediated histone deacetylation. Here, we demonstrate that ETT binds auxin directly and that this interaction determines expression of ETT target genes. However, ETT is an unusual ARF lacking the Aux/IAA-interacting Phox/Bem1 (PB1) domain ( Simonini et al., 2016 Sessions et al., 1997) and is therefore unlikely to mediate auxin signalling via the canonical pathway. This mechanism mediates precise polarity switches during organ initiation and patterning and includes the ARF, ETTIN (ETT/ARF3) as a pivotal component. We recently identified an alternative auxin-signalling mechanism whereby auxin directly affects the activity of a transcription factor (TF) complex towards its downstream targets ( Simonini et al., 2016 Simonini et al., 2017). 
This is realised by interaction with Aux/IAA repressors, leading to their ubiquitination and subsequent degradation by the 26S proteasome, ultimately relieving the repression of ARF-targeted loci ( Kelley and Estelle, 2012 Leyser, 2018 Weijers and Wagner, 2016). The main role of this auxin perception mechanism, besides non-transcriptionally inhibiting growth ( Fendrych et al., 2018 Gallei et al., 2019), is transcriptional reprogramming. As auxin levels increase, the auxin molecule binds to members of the TIR1/AFB family of auxin co-receptors ( Kepinski and Leyser, 2005 Dharmasiri et al., 2005). In canonical auxin signalling, auxin-responsive genes are repressed when auxin levels are low by Aux/IAA transcriptional repressors that interact with DNA-bound Auxin Response Factors (ARFs). In plants, this coordination can be facilitated by phytohormones such as auxin, which controls processes throughout plant development ( Vanneste and Friml, 2009). Changes in gene expression are often controlled by mobile signals that translate positional information into cell type-specific transcriptional outputs ( Hironaka and Morishita, 2012). Tight temporal and spatial regulation of the genes involved in these processes is essential for proper development of the organism. a fertilised oocyte) that proliferates into numerous cells ultimately differentiating to make up specialised tissues and organs. 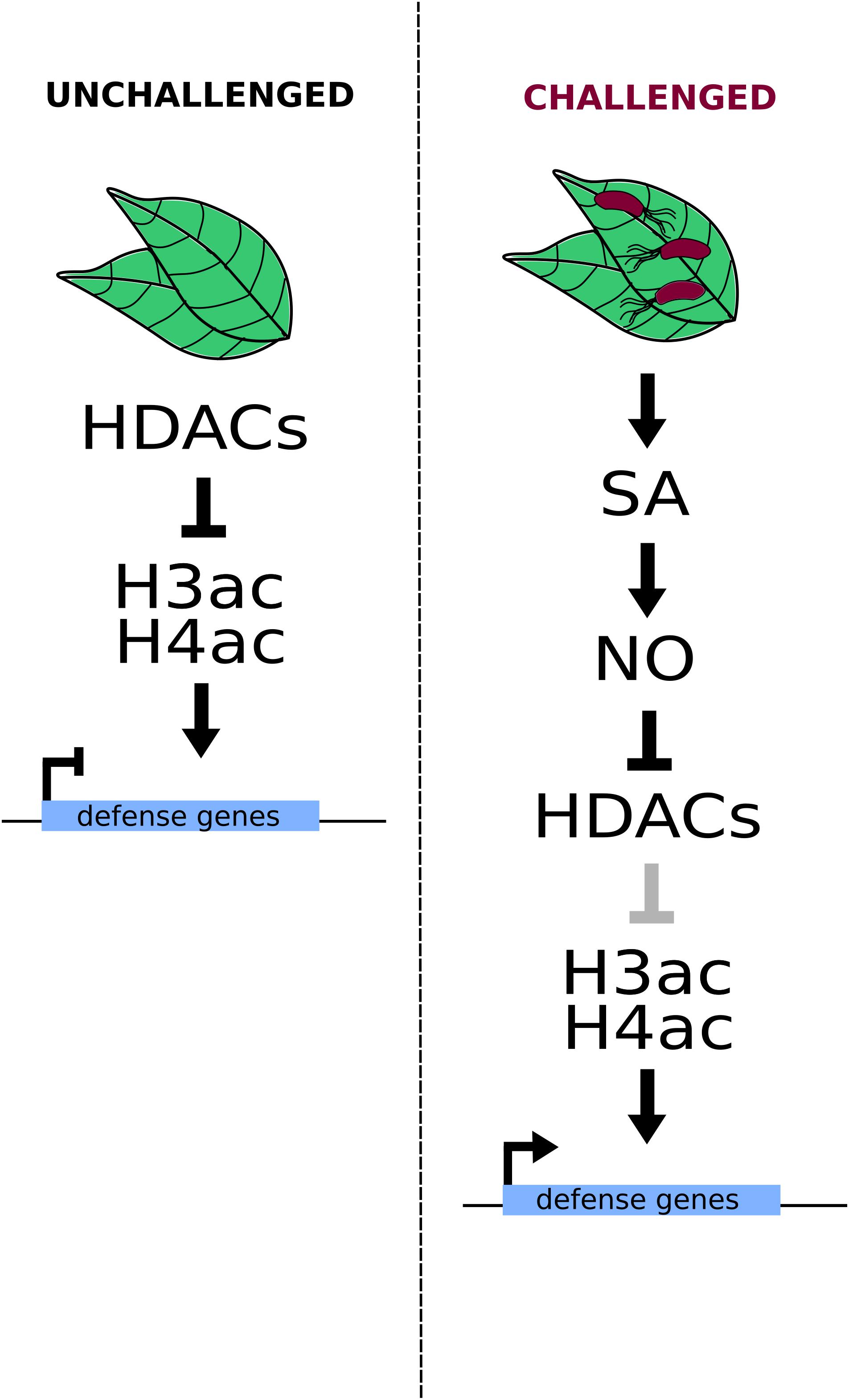
Introductionĭevelopmental programmes within multicellular organisms originate from a single cell ( i.e. Whilst auxin affects canonical ARFs indirectly by facilitating degradation of Aux/IAA repressors, direct ETTIN-auxin interactions allow switching between repressive and de-repressive chromatin states in an instantly-reversible manner. This mechanism is reminiscent of animal hormone signalling as it affects the activity towards regulation of target genes and provides the first example of a DNA-bound hormone receptor in plants. Here we show that ETTIN directly binds auxin, leading to dissociation from co-repressor proteins of the TOPLESS/TOPLESS-RELATED family followed by histone acetylation and induction of gene expression. Recently, we uncovered a non-canonical signalling mechanism for the plant hormone auxin whereby auxin directly affects the activity of the atypical auxin response factor (ARF), ETTIN towards target genes without the requirement for protein degradation. In contrast, plant hormone signalling is most commonly based on de-repression via the degradation of transcriptional repressors. Hormonal signalling in animals often involves direct transcription factor-hormone interactions that modulate gene expression.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
